
How did you get involved with the MMRF?
I was diagnosed in July 2018 with multiple myeloma. After a few office visits, I learned about the organization offering support and research information concerning this dreaded illness.
Why did you choose to participate in the MMRF Walk/Run?
It was incumbent upon me to do all that I could do within my power to help support others dealing with myeloma. The Walk/Run felt personal and was a small but important contribution from me.
The Spirit of Hope is given to “individuals/groups who inspire hope and show extraordinary commitment to the MMRF.” What does being given the award mean to you?
To say that I am honored is truly an understatement. When I was approached about this, I became extremely emotional that the team would think of me or see my journey and felt it was worthy of sharing. This means the world to me. While I was going through my chemo treatment, I would try to talk to and encourage people that I met in the waiting area or in the lab. Being given this award means I am accountable. I am accountable because I have been given the opportunity to live, to enjoy my family, to enjoy life, to work, to play, and to vacation. I feel that it is my duty to make sure that the medicines, services, tools, and information that were used for my benefit are shared by me with others.

How have you found perseverance in light of obstacles? Please share any stories that have given you strength.
I had no idea what this trial and treatment were going to look like. I was extremely nervous. But being a woman of faith, I trust God implicitly! He was a major part of my healing process. Reading my Bible, taking walks, and having a dedicated support system are all part of the journey. It is truly important to have strong people in your corner who will help pull you up when you don’t feel like getting up. Once up, you can take one step, and then two, to propel you forward. Getting out and feeling the gentle breeze and the sun on your face can be a game changer because it makes the journey seem a little lighter; it does not go away. It just becomes manageable.

Do you have a favorite mantra, quote, or lyric that gives you strength?
Yes! My favorite mantra throughout my journey and still today is, “ALL IS WELL!” maintaining a cheerful outlook is key. Keeping your mind healthy helps to keep your body healthy as well.
Anything to add?
I would like to give a heartfelt thank you to my entire Atrium clinical staff, starting with Doctor Atrash, Claire, Sydney, the lab, and a host of nurses and MA’s on the fourth floor at Levine Cancer Center that helped get me through my journey. I am here and able to celebrate this moment because of all of you.

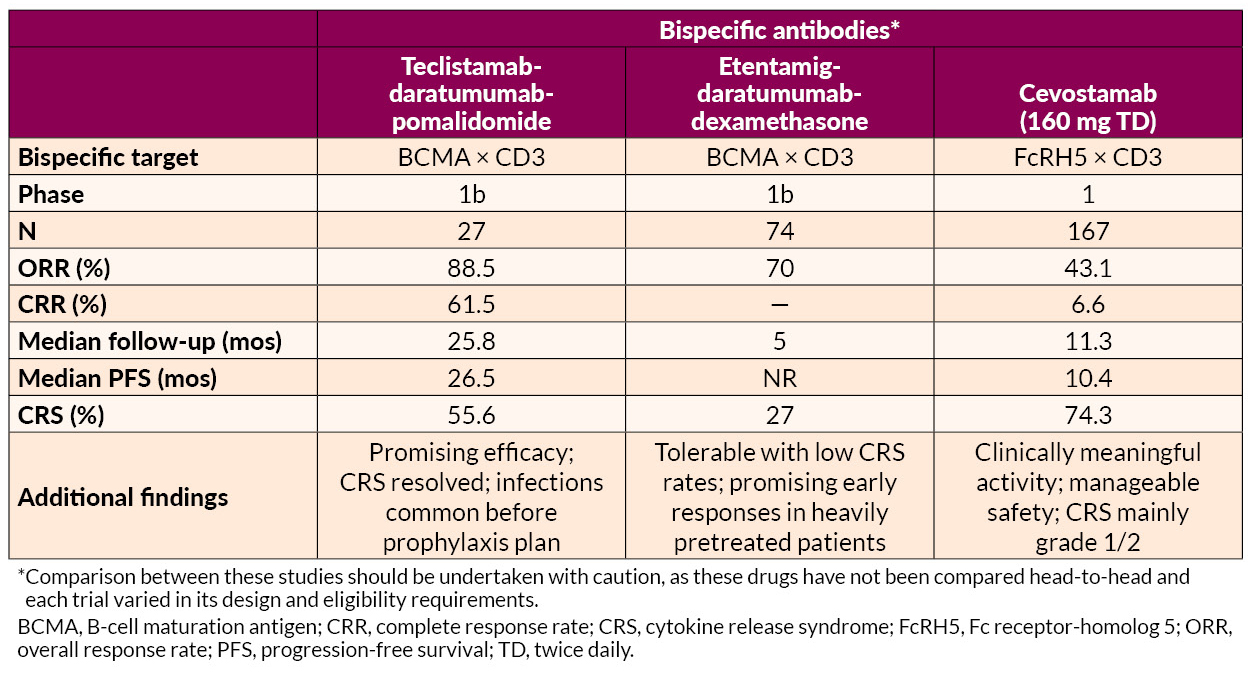
Several presentations at the 2024 American Society of Hematology (ASH) Annual Meeting relayed new and updated study data in multiple myeloma (MM), along with significant advancements in the treatment of
- Smoldering multiple myeloma (SMM)
- Prognostic indicators
- Newly diagnosed MM (NDMM)
- Maintenance therapy
- Minimal residual disease (MRD) testing
- Relapsed/refractory MM (RRMM)
- AL amyloidosis
Advances in SMM Management
Ongoing research of SMM treatment is focused on early intervention and observation with monitoring, identification of patients at high risk of progression, and novel treatment approaches. Two studies addressed these issues.
Phase 2 EMN15/HOVON147 Study: KRd vs Rd
- Intervention: Lenalidomide and dexamethasone (Rd) ± carfilzomib (K) as induction therapy
- Patient population: 58 patients with high-risk SMM (HR-SMM) identified by MAYO and/or PETHEMA* criteria
- Efficacy: At a median follow-up of 34 months, KRd significantly improved
- MRD negativity, next-generation flow cytometry [NGF]; 10-5 (57% vs 5%; odds ratio [OR]=0.04, 95% confidence interval [CI], 0.00–0.28; P<0.001)
- Progression-free survival (PFS) (hazard ratio [HR]=0.08, 95% CI, 0.02–0.35; P<0.001)
3-year PFS of 94% vs 40%. MRD negativity after 4 cycles was not significantly better with KRd (37% vs 14%; OR=0.27, 95% CI 0.06–1.12; P=0.07).
- Safety: Although KRd was associated with higher rates of grade 3/4 adverse events (AEs) (74% vs 53%), the combination resulted in fewer treatment discontinuations due to progression (6% vs 36%)
- Takeaway: The authors concluded that the addition of K as induction is “feasible and safe in patients with HR-SMM.” It is important to note that treatment of HR-SMM is still an evolving area where high-risk patients are not predictably identified; the benefit of treatment should outweigh its risks
*MAYO criteria: bone marrow plasma cells ≥10%, serum M protein ≥3 g/dL, and serum free light-chain ratio <0.125 or >8; PETHEMA criteria: presence of ≥95% abnormal plasma cells (presence or absence of CD38, CD56, CD19, and/or CD45), and immunoparesis, defined as a reduction (below the lower normal limit) in the levels of 1 or 2 of the uninvolved immunoglobulins.
Phase 3 AQUILA Study of Daratumumab
- Intervention: Daratumumab vs monitoring
- Patient population: HR-SMM (defined as clonal bone marrow plasma cells [BMPCs] ≥10% and ≥1 risk factor [serum M protein ≥30 g/L, IgA SMM, immunoparesis with reduction of 2 uninvolved Ig isotypes, serum involved:uninvolved free light chain ratio ≥8 and <100, and/or clonal BMPCs >50% to <60%])
- Efficacy: At a median follow-up of 65.2 months,
- PFS was significantly improved with daratumumab (HR=0.49; 95% CI, 0.36–0.67; P<0.0001), with a median PFS not reached in the daratumumab group vs 41.5 months in the active monitoring group
- The overall response rate (ORR) was significantly higher with daratumumab (63.4% vs 2.0%; P<0.0001), with a prolonged median time to first-line MM treatment (not reached vs 50.2 months; HR=0.46; 95% CI, 0.33–0.62; P<0.0001)
- Safety: Grade 3/4 AEs were more common with daratumumab (40.4% vs 30.1%), but the treatment was generally well tolerated, with low discontinuation rates (5.7%). The most common AE was hypertension (5.7% vs 4.6%).
- Takeaway: The authors noted that these findings “strongly support the benefit of early intervention with daratumumab monotherapy vs active monitoring.”
Functional Risk and the Role of 24-Hour Urine Tests
Two studies highlighted important aspects of prognostic indicators: one addressed functional high-risk disease status and the other assessed the value of routine 24-hour urine assessments.
Functional High Risk
Functional high-risk status could be a useful tool for identifying patients with poor prognosis, according to one analysis. Functional high-risk MM, characterized by disease progression or death within a year of initial treatment, is a poor prognostic subset regardless of high-risk cytogenetic features at diagnosis. Of the 228 functional high-risk patients identified from the Multiple Myeloma Research Foundation (MMRF) CoMMpass study, 165 were categorized as high risk and 63 as standard-risk (ie, patients with or without high-risk features [t(4;14), t(14;16), t(14;20), 1q amplification (amp1q), 17p deletion (del17p), TP53 mutations, or ISS-stage III disease]). The median overall survival (OS) was 13.2 months for the standard-risk group and 11.6 months for the high-risk group (P=0.29). Primary refractory disease was prevalent in both groups, with OS significantly shorter for patients with high-risk vs standard-risk disease (5.0 vs 7.8 months; P=0.048).
Despite differences in second-line treatment patterns (standard-risk patients received more triplet regimens; high-risk patients relied on doublets), response rates were similar, with median OS for relapsed patients 27.1 months vs 20.2 months (P=0.22 standard- vs high-risk). Overall, functional high-risk patients had an OS <24 months regardless of the presence of high-risk features at diagnosis.
This study confirms that many high-risk disease features do not present with other known high-risk features at diagnosis. More research into reliably identifying patients with high-risk features is needed.
Urine Testing
A secondary analysis of the BMT CTN 0702 (STaMINA) trial evaluating whether 24-hour urine testing adds value to MM response assessments based on International Myeloma Working Group (IMWG) criteria found that removing 24-hour urine testing requirements from response criteria resulted in changes to <1% of patient responses, with no impact on PFS prediction at any response depth. Median PFS for traditional and urine-free criteria was identical across response categories, including 63.0 months for complete response (CR) and 49.6 months for very good partial response (VGPR). Urine-free IMWG criteria remained highly prognostic for PFS (P=0.006; HR=1.05–1.36).
The authors concluded that urine-free criteria “reduce time, toxicity, and discomfort for patients, particularly those with limited dexterity,” though they also noted that these assessments remain critical in specific cases, such as AL amyloidosis or urine-only measurable disease.
Advances in NDMM Induction Therapy
Several studies highlighted the evolution of induction therapy for improving survival and quality of life for patients with NDMM. Current standard-of-care (SOC) regimens, such as quadruplet therapy incorporating daratumumab or isatuximab with bortezomib-lenalidomide-dexamethasone (VRd), continue to demonstrate superior MRD negativity and PFS regardless of autologous stem cell transplant (ASCT) eligibility, making these regimens an appropriate option for a majority of patients. Data on additional investigational therapies, including teclistamab- and belantamab-based regimens, were also presented.
Phase 3 GMMG-HD7 trial: Isatuximab-VRd
- Intervention: VRd ± isatuximab as induction therapy
- Patient population: Transplant-eligible (TE) NDMM patients
- Efficacy: After a median follow-up of 47 months, compared with VRd alone, isa-VRd:
- Significantly improved PFS (HR=0.70, 95% CI, 0.52–0.94; P=0.0184), with 3-year PFS rates of 83% vs 75%, respectively
- Provided consistent PFS benefits across most baseline characteristics through subgroup analyses, though patients with high-risk cytogenetics and poor performance status showed no significant improvement. Multivariable analysis indicated a benefit for isa-VRd (HR=0.64, 95% CI, 0.47–0.86, P=0.004)
- Takeaway: The MRD negativity benefit with isa-VRd is consistent with previous reports
Phase 3 IMROZ trial: Isatuximab-VRd
- Intervention: VRd ± isatuximab
- Patient population: Transplant-ineligible (TI) NDMM patients
- Efficacy: Compared to VRd alone, isa-VRd
- Achieved higher rates of MRD-negative CR (56% vs 41%; P=0.003; next-generation sequencing [NGS], 10-5) and sustained MRD negativity for ≥12 months (47% vs 24%)
- Had shorter median time to MRD negativity (14.7 months vs 32.8 months)
- Demonstrated greater depth of MRD negativity and more frequent positive-to-negative MRD conversions during maintenance over 36 months, correlating with PFS benefits
- Takeaway: These findings support the benefit of adding isatuximab to VRd as initiation therapy, as well as during maintenance in TI NDMM patients
Investigational Quadruplets
Notable findings regarding investigational quadruplets in NDMM were reported from two studies.
The phase 2 MajesTEC-5 study evaluated the use of teclistamab, a B-cell maturation antigen (BCMA) × CD3 bispecific antibody, in combination with daratumumab-lenalidomide-dexamethasone (DRd) or D-VRd as induction therapy for patients with TE NDMM. Out of 49 patients, all assessable patients (n=35) achieved MRD negativity (NGF, 10⁻⁵) after completing 3 treatment cycles, with sustained MRD negativity in those completing 6 cycles. Despite a high rate of cytokine release syndrome (CRS) events (65.3%, all grade 1/2), no cases of neurotoxicity or treatment discontinuation due to AEs were reported. The regimens demonstrated robust clinical efficacy with a manageable safety profile, suggesting their potential for improving long-term outcomes in this setting.
Promising results were reported from the phase 1 DREAMM-9 trial of belantamab mafodotin (belamaf) combined with VRd in TI NDMM patients. Across 8 dosing cohorts involving 108 patients, ORR was high, ranging from 71% to 100%, with the CR rate reaching 92% in certain cohorts. MRD negativity (NGS, 10-5) was most notable in higher-dose groups, achieving rates up to 75% in patients with CRs. Ocular events were the most common AE, with grade 3+ keratopathy and visual acuity scale AEs affecting up to 92% of patients in higher-dose groups, though longer dosing intervals reduced the frequency and delayed the onset of these events.
Maintenance Therapy
Also reported at ASH 2024 were findings from two studies on the use of maintenance therapy in MM, including duration of maintenance therapy, role of combination therapies vs single-agent maintenance, MRD status for informing therapy decisions, and the role of novel agents.
Phase 3 AURIGA Trial: Daratumumab + Lenalidomide vs Lenalidomide Alone
- Intervention: Lenalidomide ± daratumumab as maintenance therapy
- Patient population: TE NDMM patients who were MRD-positive after ASCT
- Efficacy: The addition of daratumumab significantly increased the MRD-negative conversion rate (10-5) at 12 months across subgroups, including patients <65 years (49.2% vs 19.7%; OR=3.95, 95% CI, 1.76–8.85) and ≥65 years (52.6% vs 17.5%; OR=5.24, 95% CI, 1.86–14.74), as well as in high-risk cytogenetic subgroups (eg, 43.8% vs 13.3%; OR=5.06, 95% CI, 1.43–17.88). PFS also favored D-R
- Safety: Grade 3/4 treatment-emergent AEs were more frequent with D-R than R, especially in Black patients (75.0% vs 66.7%) and patients <65 years (76.3% vs 63.8%)
- Takeaway: Adding daratumumab to lenalidomide maintenance improves outcomes for NDMM MRD+ patients following ASCT
Teclistamab + Lenalidomide vs Teclistamab vs Lenalidomide
- Intervention: A phase 3 safety run-in study of teclistamab combined with lenalidomide vs teclistamab vs lenalidomide maintenance therapy alone
- Patient population: NDMM patients post ASCT
- Efficacy: 100% MRD-negative CR rate at 12 months in Cohort 1, with all MRD-positive patients achieving MRD-negative status during treatment. Teclistamab was stopped after 13 cycles of treatment if ≥CR was achieved
- Safety: Grade 3/4 neutropenia and infections were common. Neutropenia occurred less frequently with reduced teclistamab dosing schedules; infections followed a similar trend: CRS occurred in 43.6% of patients but was mild (6.4% grade 2; no severe cases), and no immune effector cell-associated neurotoxicity syndrome (ICANS) was reported.
- Takeaway: Teclistamab alone or in combination with lenalidmoide can be safely administered as maintenance therapy following ASCT in NDMM
MRD Testing
MRD and Treatment Cessation
A prospective study evaluated whether discontinuing lenalidomide maintenance therapy after 3 years of sustained MRD negativity (NGF) might be feasible and safe in NDMM patients following ASCT. MRD status was assessed in patients who had achieved stringent CR and then at 6, 12, 24, and 36 months after the initiation of lenalidomide maintenance. Patients who had at ≥3 consecutive MRD-negative results and had received at least 36 months of maintenance discontinued lenalidomide maintenance only if they had also achieved imaging MRD negativity (via PET/CT scan). MRD was performed every 6 months thereafter. At 3 years, 26.3% of patients (N=194) achieved sustained MRD negativity in bone marrow and imaging and discontinued maintenance. Over a median follow-up of 32 months after discontinuation, 96% of these patients remained MRD negative at 6 months, with a gradual decline to 86% at 3 years. Median PFS for this cohort was 74 months (95% CI, 38–104 months).
According to the authors, “sustained MRD negativity after ASCT and completion of 3 years of lenalidomide maintenance may guide the safe discontinuation of maintenance” They also noted, however, that further validation in randomized trials is needed.
MRD After Induction
Several studies demonstrated that sustained MRD negativity with maintenance therapy is associated with improved outcomes. Whether TE or TI, patients showing deeper responses to therapy had prolonged PFS and OS, regardless of therapeutic approach.
- Study: Phase 3 CEPHEUS trial
- Intervention: VRd ± daratumumab in NDMM patients ineligible for or deferring transplant
- Efficacy: At a median follow-up of 58.7 months, compared to VRd alone, dara-VRD
- Improved MRD negativity rates (NGS, 10-5), 60.9% vs 39.4% (OR=2.37; 95% CI, 1.58–3.55; P<0.0001)
- Improved sustained MRD negativity at ≥12 months, 48.7% vs 26.3% (OR, 2.63; 95% CI, 1.73–4.00; P<0.0001)
- Was associated with over 80% of patients remaining progression-free at 54 months
- Takeaway: D-VRd is a potential new SOC for TI NDMM patients
Results from another study suggested that, in NDMM patients treated with quadruplet therapy and ASCT, MRD progression (defined as a ≥1-log10 increase in MRD burden) was associated with a median time of 10.1 months to IMWG-defined progression.
- 78% of patients (N=216) achieved MRD negativity at the 10⁻⁵ threshold (NGS), with 56% ceasing therapy upon confirmed negativity
- MRD progression was identified as an indicator of imminent progression, yielding a 1-year survival free of second-line failure rate of 55.6%, compared to 35% for progression not preceded by MRD progression
- Notably, MRD progression outcomes, including a 2-year OS rate of 78%, were comparable to IMWG-defined progression
The authors concluded that MRD progression challenges reliance on paraprotein-based progression criteria, suggesting the need for earlier intervention and therapies with novel mechanisms of action.
- Study: Phase 3 GMMG-HD7 trial
- Intervention: VRd ± isatuximab
- Patient population: TE NDMM patients
- Efficacy: Compared with VRd alone, isa-VRd resulted in
- Significantly higher MRD negativity rates post induction (55% vs 41%), and continued MRD negativity further improved PFS (HR=0.41, 95% CI, 0.25–0.65; P<0.001)
- Significantly longer PFS in MRD-positive patients (HR=0.64, 95% CI, 0.43–0.96; P=0.03)
- Takeaway: Adding isatuximab to standard VRd therapy achieves deeper responses and improves outcomes in NDMM
An analysis of the GEM2017FIT trial demonstrated that peripheral residual disease (PRD) assessed by mass spectrometry and MRD assessed by NGF have significant prognostic value for PFS in older TI NDMM patients.
- Both PRD and MRD negativity were strongly associated with improved PFS (PRD: HR=29, P<0.0001; MRD: HR=0.31, P<0.0001), with concordant results in 79.6% of cases
- Patients with negative PRD or MRD had superior outcomes across different fitness (Geriatric Assessment in Hematology scale ≤20 or >20) and cytogenetic risk categories, with HR PRD+/MRD+ patients achieving worse outcomes (HR=12, P<0.0001)
According to the researchers, these findings support the integration of PRD evaluation into routine clinical practice to complement MRD, improving risk stratification and informing treatment strategies for older MM patients. Taken together, these findings highlight the role of PRD and MRD negativity as a robust prognostic marker and treatment goal in NDMM.
Biomarkers and Sequencing Strategies for Relapsed MM
Several studies suggested that pretreatment biomarkers and treatment sequencing are important considerations for optimizing therapeutic responses and minimizing toxicity in relapsed MM.
Prior ASCT for Chimeric Antigen Receptor (CAR) T
MM patients had significantly shorter PFS with BCMA-directed CAR T-cell therapy if they had previously received an ASCT, according to one report. A study involving 104 patients indicated that although use of HDM/ASCT did not influence the CR rate (P=0.42), patients treated with prior HDM/ASCT experienced a shorter median PFS with CAR T therapy (9.5 vs 21 months; P=0.01). Multivariate analysis confirmed this association (HR=2.17, 95% CI, 1.25–3.74; P=0.006), independent of other factors such as high-risk cytogenetics or prior treatments. Notably, the timing between HDM/ASCT and CAR T did not influence PFS, and prior HDM/ASCT had no effect on OS, CRS, or ICANS. The authors conclude that these findings may help inform treatment sequencing in MM to optimize outcomes.
Toxicity and Durable Response to CAR T-Cell Therapy
Factors influencing toxicity and durable responses were identified in a comprehensive analysis of pretreatment biomarkers in 108 patients receiving idecabtagene vicleucel (ide-cel) for RRMM. High inflammatory markers (eg, ferritin, IL-6, IL-15; P<0.05) at baseline and elevated plasma cell burden (≥50%; P=0.02) were associated with a higher risk of ICANS, whereas higher cell doses correlated with increased CRS severity (median dose 440 vs 411 × 10⁶; P=0.01). Durable responses (defined as PFS at ≥9 months) were linked to favorable bone marrow profiles, including higher CD4:CD8 ratios and increased cytotoxic natural killer and central memory CD8+ T cells. Conversely, nondurable responses were associated with elevated inflammatory markers, prior BCMA exposure, and high levels of myeloid-derived suppressor cells (P=0.0025). Given these findings, pretreatment evaluation may help optimize patient selection and improve outcomes with ide-cel therapy.
Prior BCMA Exposure for Bispecifics
Findings suggest that response to teclistamab may depend on the timing and type of prior BCMA-directed therapy, offering insights into treatment sequencing in RRMM. In a multicenter study evaluating teclistamab in RRMM, prior exposure to BCMA-directed therapy was associated with a lower ORR (51.4% vs 61.5%; P=0.012) and shorter PFS (median 4.6 vs 8.2 months; P=0.017) than was seen in patients not previously treated with BCMA-directed therapies. Although prior BCMA-directed therapy was not independently predictive of PFS (HR=1.25, 95% CI, 0.95–1.64; P=0.1), waiting >8.7 months between BCMA therapies was linked to superior PFS (8.1 vs 2.5 months; P=0.001). Toxicity profiles were comparable between groups, but grade 3 thrombocytopenia occurred more frequently in the BCMA-exposed cohort (10.7% vs 6.6%; P=0.08).
Early RRMM: Innovations and Emerging Standards
Several studies on the treatment of early RRMM were presented. Belantamab mafodotin–based regimens demonstrated superior PFS to and deeper responses than daratumumab-based combinations, offering a potential new option for first-relapse treatment.
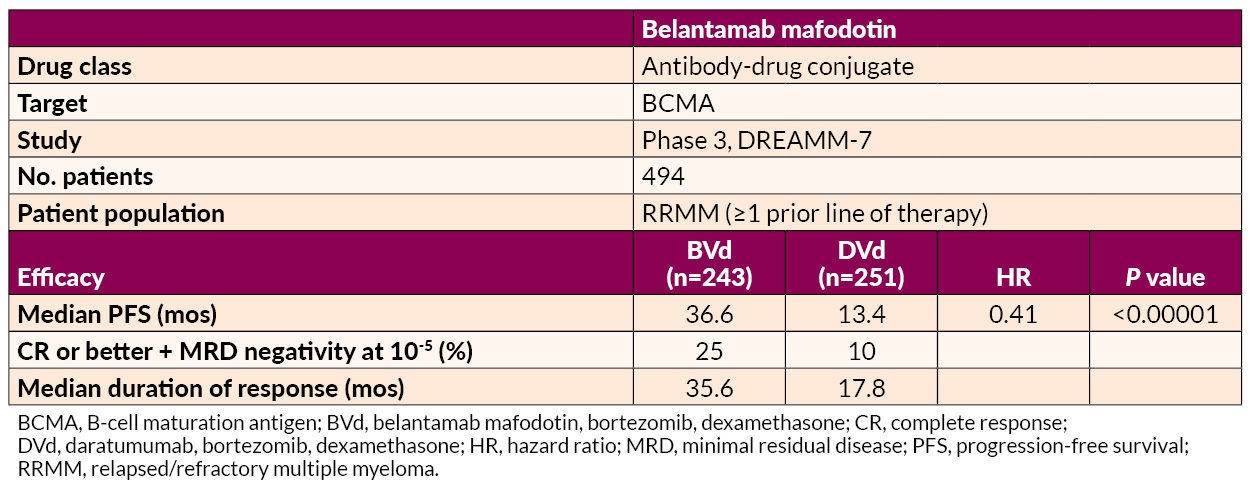
Ciltacabtagene autoleucel (cilta-cel) delivered significant benefits in MRD negativity and prolonged PFS relative to SOC in lenalidomide-refractory patients.
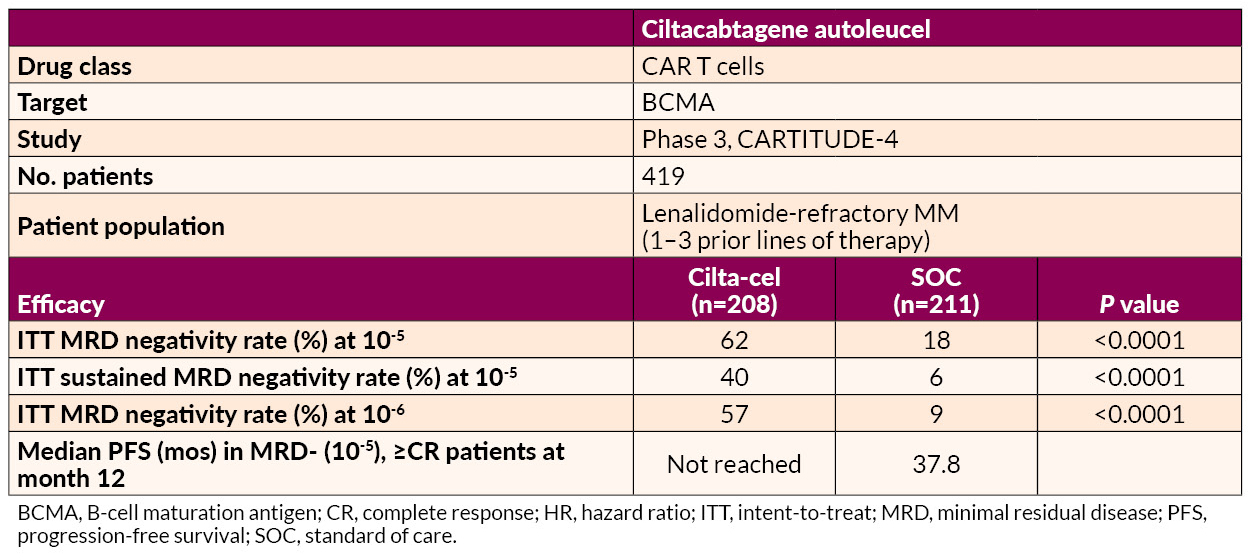
Cereblon E3 Ligase Modulator (CELMoD) and Elranatamab
Mezigdomide in combination with bortezomib or carfilzomib achieved high response rates (up to 85.7%) and durable responses (median PFS up to 17.5 months) across dose-expansion cohorts. Mezigdomide in other novel combinations, such as with agents targeting key oncogenic pathways (eg, MEK and BET inhibitors), displayed promising efficacy with manageable toxicity profiles.
Elranatamab combined with carfilzomib and dexamethasone demonstrated promising efficacy with a 100% ORR and manageable safety signals, including no dose-limiting toxicities in early-phase trials.
Innovative Approaches in Late RRMM Treatment
Several novel approaches to late treatment of RRMM with CAR T cells, including BCMA-targeted anitocabtagene autoleucel (anito-cel) and HBI0101 and bispecific antibodies, also showed promise.
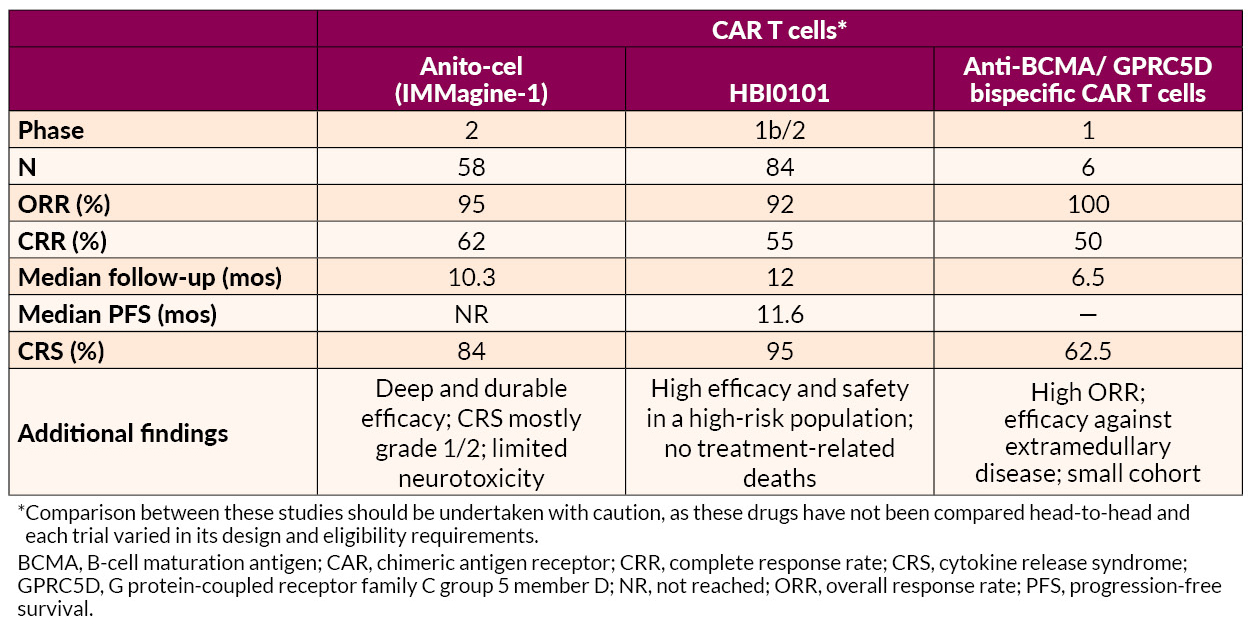
AL Amyloidosis Advances
AL amyloidosis is a plasma cell disorder and occurs in 10% to 15% of MM patients. Studies presented on AL amyloidosis showed benefits for novel drug combinations and CAR T-cell therapy. In addition, one analysis suggested that further development of therapies targeting deposited amyloid fibrils may be useful for addressing cardiac dysfunction associated with AL amyloidosis.
Phase 3 ANDROMEDA trial
- Intervention: Bortezomib + cyclophosphamide + dexamethasone (VCd) ± daratumumab
- Patient population: Newly diagnosed AL amyloidosis patients
- Efficacy: Adding daratumumab to VCd resulted in
- Higher hematologic CR rates (59.5% vs 19.2%; OR=6.03, 95% CI, 3.80–9.58; P<0.0001)
- Prolonged major organ deterioration PFS (MOD-PFS; HR=0.44, 95% CI, 0.31–0.63; P<0.0001)
- Prolonged OS (HR=0.62, 95% CI, 0.42–0.90; P=0.0121)
The median MOD-PFS was not reached for daratumumab-VCd vs 30.2 months for VCd. The 5-year survival rates were 76.1% with daratumumab-VCd vs 64.7% with VCd. Cardiac and renal responses were approximately doubled in the daratumumab-VCd group.
- Safety: Grade 3/4 AEs included lymphopenia and pneumonia
- Takeaway: According to the researchers, the findings reaffirm “this regimen’s position as the only SOC in this difficult-to-treat disease”
Phase 2 ISAMYP Study
- Intervention: Isatuximab + pomalidomide + dexamethasone (IsaPd)
- Patient population: 41 patients with relapsed or suboptimal-response AL amyloidosis
- Efficacy:
- 60% achieving ≥VGPR after 1 cycle
- 80% reaching ≥VGPR after 6 cycles
- 51% achieving CR
Treatment responses were observed early on, with a median time to initial hematologic response of 1 week and ≥VGPR within 4 weeks, including in patients with poor initial responses.
- Safety: Grade 3/4 AEs were observed in 80.5% of patients; most were manageable with dose adjustments or supportive care
- Takeaway: Rapid, deep hematologic responses were achieved with IsaPd
Phase 2 Multicenter Study for t(11;14) AL Amyloidosis
- Intervention: Venetoclax + dexamethasone (ven-D)
- Patient population: Newly diagnosed t(11;14) AL amyloidosis patients
- Efficacy:
- The CR + VGPR rate at 3 months was 58.6%, including CR in 27.6%
- The best hematologic response at any time, a composite of the CR + VGPR, was 62.9%, and the CR rate was 37.1%
- Organ responses at 6 months included improvements in cardiac function (35.0%) and renal function (90.9%)
- Safety: 3 cardiac-related deaths occurred and 6 patients discontinued due to lack of efficacy. Severe AEs such as lymphopenia and liver dysfunction affected 5.6% of patients
- Takeaway: ven-D offers rapid and high-quality hematologic responses with manageable safety risks
Another study found that the novel anti-BCMA-targeted CAR T-cell therapy HBI0101 demonstrated high efficacy and acceptable safety in 16 heavily pretreated patients with relapsed/refractory AL amyloidosis.
- Hematologic responses were present in 94%, with 75% attaining CR
- MRD negativity (flow cytometry; 10-5) was observed in 64% of evaluable patients
- Organ responses occurred in 50% of evaluable cases, with improvements noted in cardiac and renal function
- AEs included
- Grade 3/4 neutropenia (63%)
- Grade 3/4 anemia (31%)
- Manageable CRS in 88% of patients, mostly grades 1–2
The authors noted that earlier intervention before advanced cardiac disease may optimize outcomes, but HBI0101 shows potential to improve organ function and survival in this difficult-to-treat population.
Addressing Cardiac Dysfunction
Further development of therapies targeting deposited amyloid fibrils may be needed to address cardiac dysfunction in AL amyloidosis, according to one report. A single-site retrospective study of 43 patients highlighted the fact that although current systemic AL amyloidosis treatments, including daratumumab- and bortezomib-based regimens, effectively reduce toxic amyloid light chain production, they show limited impact on improving cardiac function. The study showed that 35% of patients experienced clinically significant improvement in cardiac dysfunction, as measured by global longitudinal strain (GLS), but only 10% transitioned from reduced to normal GLS. Most patients (79%) had reduced GLS at baseline, with limited meaningful changes observed across follow-up periods.
—
Jointly provided by the MMRF and RedMedEd.
Support for this activity has been provided through sponsorships from Alexion Pharmaceuticals, Inc.; Pfizer Inc.; and Sanofi US and by an educational grant from Janssen Biotech, Inc., administered by Janssen Scientific Affairs, LLC.
SOUTH SAN FRANCISCO, Calif, February 12, 2025–Opna Bio, a clinical-stage biopharmaceutical company focused on the discovery and development of novel oncology therapeutics, announced today that the FDA has granted orphan drug designation (ODD) to one of its lead programs, OPN-6602, for the treatment of multiple myeloma (MM). OPN-6602 is an oral, small molecule inhibitor of the E1A binding protein (EP300) and CREB-binding protein (CBP) currently being tested in a Phase 1 trial in patients with relapsed or refractory MM.
Read the full press release here.

How did you get involved with the MMRF?
I was introduced to MMRF through my Dallas local multiple myeloma support group.
Why did you choose to participate in the MMRF Walk/Run?
The recognition goes to my family, who surprised me by registering to participate as a group, “Team Bear” (my “grandpa” name), in the 2024 MMRF Walk/Run as a Christmas gift to me. My youngest daughter even produced a t-shirt for everyone to wear. 2024 marked my 10-year anniversary of being diagnosed, and I’m so grateful for my family’s continued support through this journey.
It is always personally beneficial to me for any research that supports myeloma. Urgency is a part of it, and there’s a possible genetic factor that could affect my children. Since myeloma affects a small percentage of people, the walk/run brings necessary attention that this disease may not usually receive.

The Spirit of Hope is given to “individuals/groups who inspire hope and show extraordinary commitment to the MMRF.” What does being given the award mean to you?
It was surprising to me to receive this award. I always think others would be much more deserving of awards than myself. I always try to support an organization that’s supporting myeloma research in any way I can. I also try to encourage attending meetings of a local support group, because it was a great resource for me and my wife when we started on this journey. I’d like to continue to benefit from them and offer the support of a local chapter to others.
How have you found perseverance in light of obstacles? Please share any stories that have given you strength.
A lot of the encouragement comes from my local support group. Due to new lines of therapy, we don’t seem to be losing as many members to the disease as they did in prior years. This gives me hope that the therapy continues to work and the options continue to expand.
I’m also encouraged because of where I live. We are blessed to live in close proximity to UT Southwestern, where one of the country’s premiere multiple myeloma research specialist practices.
Finally, my faith is what gives me the most strength. Because of my faith, I get to talk to the Guy who controls the whole universe and knows what’s going to happen to me. Regardless of the outcome, I know that He cares for me and my family. These three things, along with the support of my family, provide me hope each and every day.

Do you have a favorite mantra, quote, lyric that gives you strength?
I can’t remember where I heard this, but I like “Keep coming back, it works.” I also like what we say in our support group: “We’re sorry you’re here, but we’re glad you found us.”
Anything to add?
We are blessed to be living during a great time when myeloma is slowly becoming a chronic illness as opposed to a terminal disease. I’m thankful for all the new therapies and procedures that are being offered, with many more in the pipeline. I’m extremely appreciative of the MMRF, any support provided to myeloma research, and the help they give to the people who have the disease.
Lastly, I want to express special recognition and appreciation to Debbie, my beloved wife of 42 years, who has aggressively participated in my journey with me.

—
Learn more about the Spirit of Hope award and the 2025 recipients.

Jeff is a husband, father, and grandfather who lives with multiple myeloma every day. Despite his challenges, he faces them with optimism and a smile. Having gone through a long journey with the illness, Jeff shares his experiences, both the highs and lows, with a positive and enthusiastic attitude. His goal is to help others by offering insights from his personal battle with the disease. Jeff has participated in the MMRF Team for Cures Walk/Run Detroit since its inception and his commitment to the mission of accelerating a cure for every myeloma patient is evident in his continued efforts each year. He believes “being a 19-year survivor with a zest for life provides hope for patients” – and we agree!
How did you get involved with the MMRF?
I began to read more and visit the MMRF website after my formal diagnosis of myeloma in January 2018. I had my first solitary isolated plasmacytoma in 2006 and another in 2014. I was so impressed by my oncologist’s involvement in the Moving Mountains for Multiple Myeloma and taking patients on these adventures with him that I recognized how important the MMRF was in researching finding a cure and giving each of us hope for a positive quality of life.

Why did you choose to participate in the MMRF Walk/Run?
When it was announced that Southeast Michigan was going to have its first fundraising walk, I knew I had to participate. This will be my fourth walk. I was out of town for the second walk but my wife and I along with my daughter and son-in-law did a 5k in Cincinnati wearing our shirts. The walk became important as a vehicle to raise more awareness of myeloma, help to generate more funds for continued research, provide support for those who are in treatment, and those of us who are coping with the diagnosis, and provide hope for an opportunity to live our best lives possible post-treatment.
The Spirit of Hope is given to “individuals/groups who inspire hope and show extraordinary commitment to the MMRF.” What does being given the award mean to you?
I never thought about awards, just about fundraising and support for an organization focused exclusively on patients like me to provide continued research and clinical trials to allow patients to have a positive quality of life. And find a way to cure myeloma in the very near future. I am humbled and appreciate that MMRF recognized my contribution to the organization and my commitment to finding a way to help each of us live our best lives.

How have you found perseverance in light of obstacles? Please share any stories that have given you strength. I am a person with a positive outlook and grateful for the love of my family and friends. I have many goals in my life and their support has given me the strength to keep moving forward and appreciate each day.
I have decided to give back as a volunteer with several causes. I am a weekly volunteer at my cancer center, helping patients, nurses, and clinicians with anything they need. I am so grateful to my team for their support in getting me through the difficult times so I pay it forward. I believe being a 19-year survivor with a zest for life provides hope for patients. While they say I am inspiring, I am constantly being inspired by their will and determination to get through their diagnosis. I have met so many wonderful people and feel I was a help when I leave. I work with our social worker with anything she asks. I’ll speak with patients about my experience, listen to their concerns, and try to help in any way possible.
I also volunteer with our community food bank. So many people have food insecurity and sadly there is so much food waste. Twice a week I pick up food and deliver it to a Meals on Wheels location that cooks and sends meals out to those in need.
During the toughest part of my induction therapy, when neuropathy crippled my legs, feet, and hands and I couldn’t walk or grip silverware, I was touched by the family and friends who came from out of town (25 people, some who showed up multiple times) to help me and my wife when I was at my lowest point. Their love and support were a tremendous help in getting me back on my feet as they took me to therapy appointments and helped me do my exercises to get me back to a positive quality of life.
Do you have a favorite mantra, quote, lyric that gives you strength?
At this point in life, my mantra is to “Keep on Truckin’”, and share as many experiences as possible and memories as possible with my family. We enjoy being together and sharing good times. Music plays a big role in my life and experiencing concerts together brings me joy. I’m a huge Grateful Dead fan and we’ve been to several Dead and Company shows together. This spring my wife and family are traveling together to Las Vegas to see shows at the Sphere. We are all so excited!

Anything to add?
I really believe no one knows what tomorrow will bring. Live each day to its fullest. Do my best to be healthy. Do my Mitzvahs (good deeds), slow down, be kind. Show family and friends how special they are in your life. Be grateful and kind. If I practice these traits, I’m living my dream.
—
The MMRF is thrilled to recognize Jeff Levison as the MMRF Spirit of Hope Honoree at the 2025 MMRF Team for Cures: Detroit Walk/Run. Donate to Jeff Levinson and his team, K- Farm Guys, today to accelerate a cure!
This award is presented at every Walk/Run to a patient, caregiver, or family who inspires hope through their resilience, perseverance, and dedication to the MMRF and its mission.
Learn more about the Spirit of Hope award and the 2025 recipients.
We are deeply saddened about the passing of Richard “Dick” Parsons, a prominent and accomplished leader who held top posts at Time Warner and Citigroup among other companies. Dick was a very close friend to and long-time supporter of the Multiple Myeloma Research Foundation.
Diagnosed with multiple myeloma in 2015, Dick soon became a strong advocate for raising awareness of this uncommon blood cancer. Myeloma disproportionately affects the Black community, and Dick was an incredible partner who voiced the need for stronger data and research to drive cures. In fact, Dick participated in the MMRF CoMMpass StudySM, contributing his own data to this large longitudinal study to advance research efforts to benefit all patients. He championed the importance of taking action by learning about the disease and advocating for one’s own care, which remains an invaluable gift and legacy to the myeloma community.
Dick will be greatly missed. We extend our most heartfelt condolences to his family and loved ones.

Richard “Dick” Parsons with MMRF Founder Kathy Giusti and Sid Mukherjee.
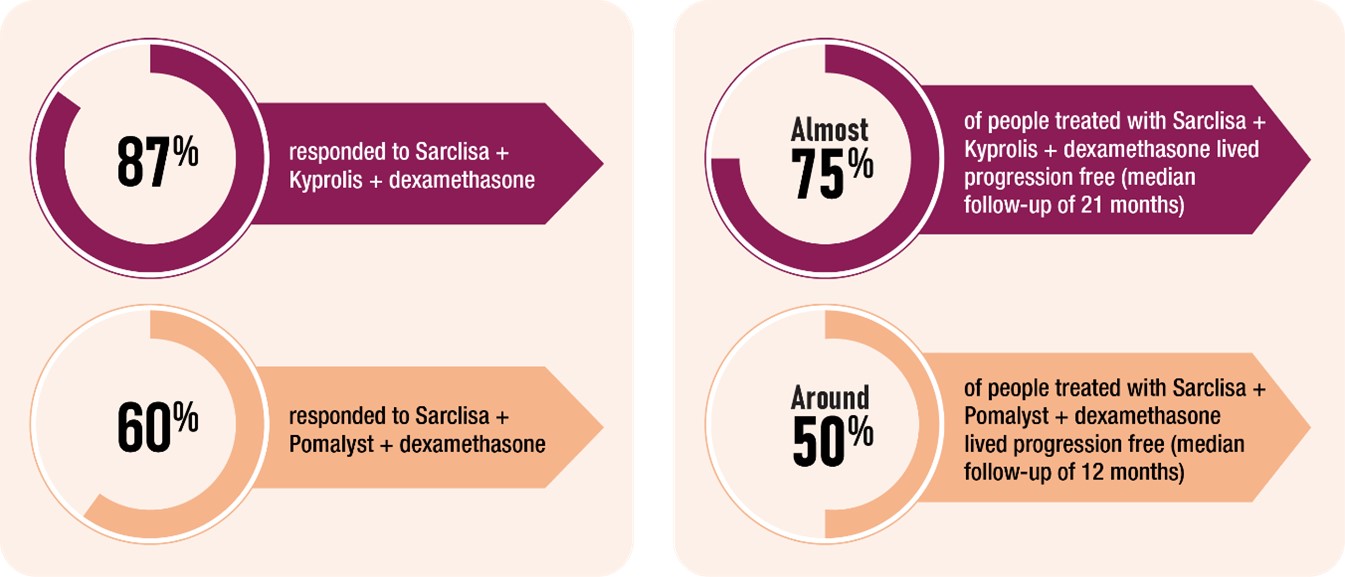
In September 2024, Sarclisa (isatuximab) became the first drug to be approved by the FDA for use with Velcade, Revlimid, and dexamethasone (Isa-VRd) for adult patients who are newly diagnosed with multiple myeloma but unable to receive high-dose chemotherapy and a stem cell transplant commonly used in myeloma treatment.
With this new approval, patients with newly diagnosed myeloma have an exciting new treatment option. Previously, Sarclisa had been approved for use as myeloma treatment, in combination with Pomalyst or Kyprolis and dexamethasone, in later lines of therapy.
In clinical trials, most patients who received a combination therapy that included Sarclisa responded to the treatment, and many lived without their myeloma getting worse.
Sarclisa is a monoclonal antibody, a type of immunotherapy agent that is designed to work with your immune system to find, bind to, and destroy myeloma cells in your body.
Importantly, Sarclisa is one of a class of agents that is able to target a specific component of myeloma cells, a protein called CD38. Myeloma treatments that target CD38, which include Darzalex (daratumumab), have several advantages, including the potential for deep responses and longer periods in which myeloma does not progress.
Patients are given Sarclisa by intravenous infusion. Treatment spans cycles of 28 days, in which Sarclisa is administered along with the other agents in the combination therapy. In the first cycle, Sarclisa is usually given weekly. For the remainder of treatment, Sarclisa is usually given every 2 weeks. Currently, studies are looking at whether giving Sarclisa as an injection (instead of an infusion) is an option.
As with most myeloma treatments, there are side effect risks with Sarclisa. Infusion reactions may occur during or shortly after the medication is given. These reactions are most common during the first infusion and can produce fever, chills, nausea, headache, difficulty breathing, low blood pressure, and flushing.
Patients receive medicines before each Sarclisa infusion to help make infusion reactions less frequent and severe. While receiving Sarclisa, patients are monitored for infusion reactions. If a reaction occurs, the infusion can be slowed down or stopped depending on the severity of the reaction.
Low white blood cell, low red blood cell, and low platelet counts occur in most patients. These effects can often be reversed by adjusting the treatment or with supportive measures like transfusions or medications to boost blood cell production. Other side effects include diarrhea, fatigue and nausea, which are typically mild and temporary. Respiratory infections, such as colds or coughs, may develop and usually improve with treatment.
Interested in learning more about Sarclisa? Talk to an MMRF patient navigator at the Patient Navigation Center at 1.888.841.6673 (Monday through Friday, 9:00 am to 7:00 pm ET) or email [email protected].
Additional Resources
MMRF Patient Toolkit: Multiple Myeloma Treatment Overview
All our downloadable resources: Education Resource Hub
The MMRF aims to incorporate a variety of new bispecific antibodies, cell therapies, checkpoint inhibitors and novel small molecules into the Horizon study.
This article presents a Q&A with MMRF Patient Navigation Center nurses Grace Allison and Brittany Hartmann on key takeaways from the 2024 annual meetings of the National Comprehensive Cancer Network (NCCN) and the Journal of the Advanced Practitioner in Oncology (JADPRO) on recent multiple myeloma treatment advances.
What have we learned about the side effects of FDA-approved CAR T-cell therapies and how they can be managed?
Low white blood cell, red blood cell, and platelet counts commonly occur with many different myeloma therapies, and they are seen even more frequently in patients receiving CAR T-cell therapy.
Because low white blood cell counts increase the risk of infections, medications called growth factors (for example, filgrastim) may be given to patients who receive CAR T-cell therapy. These drugs help the body produce white blood cells. Low red blood cell counts can cause fatigue and shortness of breath and are usually treated with blood transfusions or drugs (for example, erythropoiesis-stimulating agents) to help the body make more red blood cells. For low platelets, which raise the risk of bleeding and bruising, platelet transfusions may be necessary in severe cases.
Infections are also a common side effect of CAR T-cell therapy. Before starting treatment, patients are usually given medications to prevent infections—for example, acyclovir to prevent viral infections. Antibiotics and antifungals are commonly given, as well. Intravenous immunoglobulin (IVIG), which boosts a patient’s immune system following CAR T-cell therapy, is also often given. As another strategy for infection prevention, many treatment centers are re-vaccinating patients following CAR T-cell therapy.
If a fever develops after CAR T-cell therapy, even if it’s just low grade, patients should immediately report it to their care teams. The doctor will investigate to see if it’s caused by an infection. This may require blood tests and possibly a chest x-ray to rule out the possibility of an active infection.
If there’s no sign of infection, the doctor will see if the fever is caused by a reaction called cytokine release syndrome or CRS. In CRS, the body’s immune system gets revved up too high, resulting in an overreaction that can cause fever, chills, fatigue, muscle aches, nausea, and a fast heartbeat. In more serious cases, CRS can cause problems with the lungs, liver, or heart. Fortunately, CRS is a condition that the care team will be ready to address. Treatments for CRS include tocilizumab and steroids.
Side effects that are of particular concern are lesser-known neurotoxicities, including a condition called immune effector cell-associated neurotoxicity syndrome—better known as ICANS. In ICANS, inflammation interferes with the brain, leading to symptoms like confusion, difficulty speaking, headaches, and feeling very sleepy. In more serious cases, seizures or changes in alertness can occur.
ICANS typically occurs within a few days of receiving CAR T-cell therapy. The duration varies, but the condition usually resolves within 1 to 2 weeks with appropriate management. To manage ICANS and other neurotoxicities, the care team will usually consult with a neurologist. Management strategies can include using anti-seizure medications.
Another concern is more serious Parkinsonian-like syndromes, which are conditions that cause symptoms like those seen in Parkinson’s disease, such as stiffness, tremors, and balance problems. In studies, some patients who received the CAR T-cell agent Carvykti developed Parkinsonian-like syndromes 40 to 60 days after treatment. The symptoms may persist, requiring long-term management and supportive care.
Three bispecific antibodies (Tecvayli, Elrexfio, and Talvey) have been approved by the FDA for myeloma. What have we learned about their side effects and how they can be managed?
As with CAR T-cell therapy, CRS and ICANS can develop in patients receiving bispecific antibody therapy. CRS is more common than ICANS, but the risk for both is much lower with bispecifics than with CAR T. When these conditions develop, it’s usually at the beginning of treatment, during the step-up doses. This is why these doses are given in a hospital or other treatment facility. After all the step-up doses have been completed, the risk of CRS and ICANS is lower.
Bispecific antibodies can weaken the immune system, increasing the risk of infections. Patients and caregivers should be vigilant for signs of infection such as fever, chills, or a persistent cough. Maintaining open communication with your healthcare team about any symptom is important.
The bispecific antibody Talvey has some unique side effects. Talvey targets a protein called GPRC5D, which is present on myeloma cells. It’s also present on other cells in the body, including those found in the mouth, skin, and nails. Because of this, Talvey can cause side effects in these parts of the body, such as a full-body rash, peeling of the skin, and changes in the fingernails. Though inconvenient and unpleasant, these side effects rarely create difficulty with daily activities and instead tend to be quality-of-life issues.
Oral side effects can include taste changes and mouth sores. Difficulty swallowing is another possible side effect and can be concerning, because it can cause lack of appetite, which can lead to weight loss. Proper nutrition is important not just to maintain day-to-day health but especially to help fight off myeloma and the side effects of myeloma treatment.
Care teams often recommend a thick barrier lotion like Aquaphor for rashes and peeling. Other products are available to help strengthen and protect the nails. For oral side effects, special mouthwashes (for example, dexamethasone) can be used for dry mouth.
Consulting a nutritionist early on is important, as a nutritionist can recommend strategies for coping with mouth problems such as mouth sores, changes in sense of taste, dry mouth, and difficulty swallowing before they become a serious issue that interferes with eating.
How can the care team balance a patient’s comorbidities with his or her treatment?
For patients with comorbidities, early referral to a nutritionist is important. Additionally, physical therapy for strength maintenance and muscle protection can help patients maintain their quality of life.
One presentation at NCCN noted that a strategy for balancing comorbidity management and myeloma treatment is a drug holiday—that is, a period where a patient does not take any medication. This is particularly an option when the myeloma is well controlled. This strategy can make patients nervous that the myeloma will come roaring back. Care teams, however, are very much on board with drug holidays when appropriate, and they don’t fear that it will affect long-term outcomes.
What other multiple myeloma treatments are being explored?
CELMoDs, a new class of myeloma drug, work like immunomodulators such as Revlimid and Pomalyst and are orally administered. They help the immune system recognize and attack myeloma cells, and they kill myeloma cells directly. CELMoDs have been shown to be effective even in patients whose myeloma has become resistant to some treatments.
One point that came up at JADPRO was the importance of making CELMoDs and oral agents more widely available—for instance, to patients being treated in community facilities. Expanding the availability of these agents would make these treatments accessible to patients who do not live near specialized treatment facilities and could also reduce or eliminate the need for patients to spend time receiving infusions. Overcoming these barriers offers the potential to improve patient quality of life while also providing treatments that have shown to have great responses.
What are the current barriers to accessing myeloma treatment? What steps are being taken to make access more equitable?
We know that the percentage of myeloma deaths is higher in Black, Latinx, and LGBTQ+ patients, largely because of unequal access to care and lower rates of early detection and screening. The fact that there is an awareness of this discrepancy, and that it’s being discussed, is an important step forward. This is an area that the MMRF is actively addressing by raising awareness among and providing education to care providers.
Other efforts to improve care equity have focused on making participation in clinical trials easier for a broader population of patients. Strategies like offering travel assistance and expense reimbursement, and opening trials in smaller community settings and not just large academic hospitals, allow patients from rural areas to participate.
Finally, helping patients manage treatment-related costs is another area that has received greater attention. This can involve helping patients navigate the ever-changing landscape of insurance or offering programs that provide financial support. Receiving CAR T-cell therapy involves a significant commitment and can disrupt daily life, including work responsibilities. At present, this treatment requires travel to a specialized medical center, which for many patients will be far from home, adding time and logistical challenges. This time away from work and daily life can be a burden, both emotionally and financially. Several organizations have developed programs to provide financial support for treatment-related expenses, such as the cost of medication, travel to a treatment center, and hotels.
HOUSTON & SEATTLE, December 17, 2024–Indapta Therapeutics, Inc., a privately held clinical stage biotechnology company developing next-generation cell therapies for the treatment of cancer and autoimmune diseases, announced today it has closed a $22.5 million round of new financing to accelerate the clinical development of its differentiated allogeneic Natural Killer (NK) cell therapy. Current investors RA Capital Management, LP, Leaps by Bayer, the impact investment arm of Bayer AG, Vertex Ventures HC, Pontifax, and the Myeloma Investment Fund, the venture philanthropy subsidiary of the Multiple Myeloma Research Foundation, completed the round.
“This funding will enable us to generate significant additional data in our ongoing trial of IDP-023 in cancer as well as initial data from our first trial in autoimmune disease,” said Mark Frohlich, Indapta’s CEO. “Preliminary results of IDP-023 in cancer are encouraging and we look forward to initiating our Phase 1 trial for multiple sclerosis in Q1 2025. This financing, together with our recently announced collaboration with Sanofi, highlights the promise of our differentiated platform.”
Read the full press release here.


